As Egerton Law recently reported the medical device manufacturer Zimmer, Inc., has issued a recall of all lots and sizes of their Persona Trabecular Metal Tibial Plate/Persona TM Tibia. The device, on the market since late 2012, was recalled after increased complaints of loosening after implantation, which sometimes requires revision surgery to correct.
Since our initial report, we have received inquiries from many individuals who have suffered pain after undergoing total knee replacement surgery and wonder if they have a claim. If you have continued problems after knee replacement surgery, and joint replacement surgery in general, you may find the following questions and answers helpful.
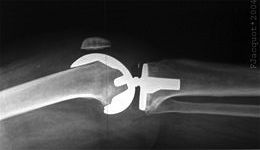
X-ray of an example total knee replacement, side view. / Wikipedia
Q: I got my knee replaced before 2012 and have had problems ever since. I know it is a Zimmer. Do I have a claim?
A: The recently recalled component of the Zimmer Persona device was not used until late 2012, so you do not have a claim related to this recall. In fact, Zimmer manufactures several different knee replacement models that have had high rates of success. However, Zimmer previously issued a recall on the NexGen MIS Tibial Components, requiring a change in the surgical technique used in connection with this device. If you received the NexGen MIS device prior to the updated surgical techniques, you may also have a claim.
Q: I know that I have a Zimmer knee replacement that was implanted after 2012. I have not been having any trouble, but I am worried I might have the recalled device. What should I do?
A: If you have no problems after receiving a knee replacement, you are part of the majority of people who experience a successful joint replacement surgery. Joint replacement devices may be recalled after a higher than normal failure rate is discovered. This does not mean that everyone who receives the affected device will experience problems. Actually, a “higher than normal” failure rate is generally still a very small percentage, and the chances that your device will fail is very low. If you do begin to experience problems down the road, make sure you follow up with your doctor and report these changes.
Q: I had a knee replacement that had to be revised, but I don’t know what company manufactured it. How do I know if I have a claim?
A: The first step in determining whether you have a claim against the manufacturer of a device is to find out which device you received. Hospital records will provide you with this information.
Other factors to be considered are what caused the need for a revision surgery (was it a problem with the device, or something else?), and how much time has passed since your original knee replacement surgery. Joint replacements typically have a life-span of 15 to 20 years. If you required revision surgery long before the expected life span, there is a possibility of premature device failure.
Q: I had my knee replaced within the last year, and it still doesn’t feel right. How do I know if I need a revision surgery?
A: Everyone has a different healing process after joint replacement surgery. Talk with your doctor if you are experiencing continued pain after a knee replacement. Make sure you follow through with all physical therapy exercises, and ask questions at doctor visits if you are unsure whether you are healing properly. You can also visit a new orthopaedic doctor for a second opinion if you choose.
We at Egerton Law hope this article helps to answer some of the more common questions about the Zimmer Persona knee replacement recall. If you have additional questions regarding your knee replacement and any potential claims, a medical device attorney can help you find answers. Visit our website for more information.
http://www.webmd.com/osteoarthritis/guide/knee-replacement-surgery?page=1#0
(info on lifespan of knee replacement & physical therapy surgery)