Recipients of the DePuy ASR metal-on-metal hip implant who have undergone revision surgery between January 31, 2015, and February 15, 2017, are finally able to participate in the Settlement Program created by the manufacturer.
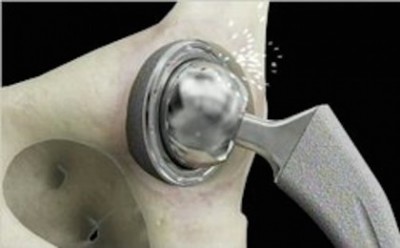
The ASR hip implant, which was recalled in 2010, was recalled by DePuy Orthopaedics after reports of high revision rates at 5 years after implantation. In 2013 the first group of injured plaintiffs was able to receive compensation by participating in the Settlement Program. Since the last deadline in 2015, individuals who had the recalled metal-on-metal hips revised have had no recourse for compensation for their injuries.
Image / Hip Replacement Survivors
While some people who received the DePuy ASR or other metal-on-metal hip devices will not experience problems, others are affected by metal ions entering the blood stream due to the contact of the metal components of the device. This effect, called metallosis, can have a variety of symptoms, including damage to the tissue surrounding the hip joint. Pain, squeaking, popping, and clicking have been reported by many patients.
In some cases the symptoms are so serious that a revision surgery is required to remove the metal-on-metal device, which is usually replaced by an implant using ceramic or plastic components.
DePuy’s other metal-on-metal hip implant, the Pinnacle Ultamet, was removed from the market in 2013 but has not been recalled. Recent trials have resulted in two large verdicts for the plaintiffs, and another trial has been scheduled for September.
Egerton Law has been representing people injured by metal-on-metal hip implants since 2010. See our website for more information.